A comment on the last post
Richard Cyganiak Says: May 19th, 2007 at 7:55 pm e
Thanks for this nice introduction Peter. Note that the DBpedia URIs also work in a web browser, so you can go to http://dbpedia.org/resource/Uppsala and the DBpedia server will generate a web page showing the information it has about the item.
I have been playing some more with dbpedia and RDF. (It is important to realise that – as in the above example – many of the accesses will be through UIs, not raw RDF, so casual readers shouldn’t worry). There have been several ways of expressing RDF and these are slightly confusing but the important thing is to know what the predicates and potential values are. The exploratory approach (using a browser) Richard suggests is useful.
Let’s look at chemistry. Assume we know every little about dbpedia but we’ll look at some chemistry. So access Arsenic in Wikipedia
Arsenic
From Wikipedia, the free encyclopedia
Categories: Articles with unsourced statements since February 2007 | All articles with unsourced statements | Metalloids | Pnictogens | Toxicology | Chemical elements | Arsenic
PMR> Note that these Categories will be reusable in RDF triples.
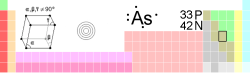
33 germanium ← arsenic → selenium P
↑
As
↓
Sb
General Name, Symbol, Number arsenic, As, 33 Chemical series metalloids Group, Period, Block 15, 4, p Appearance metallic gray

Standard atomic weight 74.92160(2) g·mol−1 Electron configuration [Ar] 3d10 4s2 4p3 Electrons per shell 2, 8, 18, 5 Physical properties Phase solid Density (near r.t.) 5.727 g·cm−3 Liquid density at m.p. 5.22 g·cm−3 Melting point 1090 K
(817 °C, 1503 °F)Boiling point subl. 887 K
(614 °C, 1137 °F)Critical temperature 1673 K Heat of fusion (gray) 24.44 kJ·mol−1 Heat of vaporization ? 34.76 kJ·mol−1 Heat capacity (25 °C) 24.64 J·mol−1·K−1
P(Pa) 1 10 100 1 k 10 k 100 k Atomic properties Crystal structure rhombohedral Oxidation states ±3, 5
(mildly acidic oxide)Electronegativity 2.18 (Pauling scale) Ionization energies
(more)1st: 947.0 kJ·mol−1 2nd: 1798 kJ·mol−1 3rd: 2735 kJ·mol−1 Atomic radius 115 pm Atomic radius (calc.) 114 pm Covalent radius 119 pm Van der Waals radius 185 pm Miscellaneous Magnetic ordering no data Electrical resistivity (20 °C) 333 n Ω·m Thermal conductivity (300 K) 50.2 W·m−1·K−1 Young’s modulus 8 GPa Bulk modulus 22 GPa Mohs hardness 3.5 Brinell hardness 1440 MPa CAS registry number 7440-38-2 Selected isotopes
iso NA half-life DM DE (MeV) DP 73As syn 80.3 d ε – 73Ge γ 0.05D, 0.01D, e – 74As syn 17.78 d ε – 74Ge β+ 0.941 74Ge γ 0.595, 0.634 – β– 1.35, 0.717 74Se 75As 100% As is stable with 42 neutrons References Arsenic (IPA: /ˈɑːsənɪk/, /ˈɑɹsənɪk/) is a chemical element that has the symbol As and atomic number 33. Its Atomic Mass is 74.92. Its Ionic Charge is (3-) Its position in the periodic table is shown at right. This is a notoriously poisonous metalloid that has many allotropic forms: yellow (molecular non-metallic) and several black and gray forms (metalloids) are a few that are seen. Three metalloidal forms of arsenic with different crystal structures are found free in nature (the minerals arsenic sensu strictu and the much rarer arsenolamprite and pararsenolamprite), but it is more commonly found as arsenide and arsenate compounds. Several hundred such mineral species are known. Arsenic and its compounds are used as pesticides, herbicides, insecticides and various alloys.
The box on the right is the “infobox” and it is many of these properties that find their way into dbpedia. The property names are not yet fully standardised and not all are memorable, but Wikipedian chemists can – and I suspect will – work on these to stnadrdaize. In which case they can be used for RDF/SPARQL queries. For example:
[… white space inserted for formatting – scroll down …]
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
.
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/resource/Category:Chemical_elements | |
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/resource/Category:Toxicology | |
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/resource/Category:Metalloids | |
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/resource/Category:Pnictogens | |
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/resource/Category:Arsenic | |
| As | http://www.w3.org/1999/02/22-rdf-syntax-ns#type | http://dbpedia.org/class/yago/chemical_element | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Articles_with_unsourced_statements | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Chemical_elements | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Toxicology | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Metalloids | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Pnictogens | |
| As | http://www.w3.org/2004/02/skos/core#subject | http://dbpedia.org/resource/Category:Arsenic | |
| As | http://dbpedia.org/property/reference | http://www.asmalldoseof.org/ | |
| As | http://dbpedia.org/property/reference | http://www.atsdr.cdc.gov/HEC/CSEM/arsenic/ | |
| As | http://dbpedia.org/property/reference | http://www.clu-in.org/contaminantfocus/default.focus/sec/arsenic/cat/Overview/ | |
| As | http://dbpedia.org/property/reference | http://www-cie.iarc.fr/htdocs/monographs/vol23/arsenic.html | |
| As | http://dbpedia.org/property/reference | http://www.greenfacts.org/arsenic/arsenic-1.htm | |
| As | http://dbpedia.org/property/reference | http://www.inchem.org/documents/ehc/ehc/ehc224.htm | |
| As | http://dbpedia.org/property/reference | http://www.npi.gov.au/database/substance-info/profiles/11.html | |
| As | http://dbpedia.org/property/reference | http://www.webelements.com/webelements/elements/text/As/index.html | |
| As | http://dbpedia.org/property/reference | http://www.origen.net/arsenic.html | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsenic | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsen | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsénico\” | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsenic | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsenico | |
| As | http://www.w3.org/2000/01/rdf-schema#label | ヒ素\” | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsenicum | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsen | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsênio\” | |
| As | http://www.w3.org/2000/01/rdf-schema#label | Arsenik | |
| As | http://www.w3.org/2000/01/rdf-schema#label | 砷\” | |
| As | http://dbpedia.org/property/number | 33 | |
| As | http://dbpedia.org/property/symbol | As | |
| As | http://dbpedia.org/property/left | http://dbpedia.org/resource/germanium | |
| As | http://dbpedia.org/property/right | http://dbpedia.org/resource/selenium | |
| As | http://dbpedia.org/property/above | http://dbpedia.org/resource/phosphorus | |
| As | http://dbpedia.org/property/below | http://dbpedia.org/resource/antimony | |
| As | http://dbpedia.org/property/color1 | #cccc99 | |
| As | http://dbpedia.org/property/color2 | black | |
| As | http://dbpedia.org/property/group | 15 | |
| As | http://dbpedia.org/property/period | 4 | |
| As | http://dbpedia.org/property/block | p | |
| As | http://dbpedia.org/property/k | [[sublimation (physics)|subl.]] 887 | |
| As | http://dbpedia.org/property/k | 1090 | |
| As | http://dbpedia.org/property/c | 614 | |
| As | http://dbpedia.org/property/c | 817 | |
| As | http://dbpedia.org/property/f | 1137 | |
| As | http://dbpedia.org/property/f | 1503 | |
| As | http://dbpedia.org/property/mn | 73 | |
| As | http://dbpedia.org/property/mn | 74 | |
| As | http://dbpedia.org/property/mn | 75 | |
| As | http://dbpedia.org/property/sym | As | |
| As | http://dbpedia.org/property/na | 100 | |
| As | http://dbpedia.org/property/na | http://dbpedia.org/resource/synthetic_radioisotope | |
| As | http://dbpedia.org/property/n | 42 | |
| As | http://dbpedia.org/property/hl | [[1 E6 s|17.78]] d | |
| As | http://dbpedia.org/property/hl | [[1 E6 s|80.3]] [[day|d]] | |
| As | http://dbpedia.org/property/dm1 | ε | |
| As | http://dbpedia.org/property/dm1 | http://dbpedia.org/resource/electron_capture | |
| As | http://dbpedia.org/property/de1 | – | |
| As | http://dbpedia.org/property/pn1 | 73 | |
| As | http://dbpedia.org/property/pn1 | 74 | |
| As | http://dbpedia.org/property/ps1 | http://dbpedia.org/resource/germanium | |
| As | http://dbpedia.org/property/dm2 | http://dbpedia.org/resource/Positron_emission | |
| As | http://dbpedia.org/property/dm2 | http://dbpedia.org/resource/Gamma_radiation | |
| As | http://dbpedia.org/property/ps2 | – | |
| As | http://dbpedia.org/property/ps2 | http://dbpedia.org/resource/germanium | |
| As | http://dbpedia.org/property/de2 | 0.05[[Delayed nuclear radiation|D]], 0.01D, [[Conversion electron|e]] | |
| As | http://dbpedia.org/property/de2 | 0.941 | |
| As | http://dbpedia.org/property/pn2 | 74 | |
| As | http://dbpedia.org/property/dm3 | γ | |
| As | http://dbpedia.org/property/de3 | 0.595, 0.634 | |
You can work out some of the property names by comparing against their values. For example…
| http://dbpedia.org/resource/Arsenic | As | http://dbpedia.org/property/c |
links to the melting point in degC. There is still some work to be done (e.g. linking properties and units) but RDF is well suited for this and we’ll be working out some approaches.
hello,
I might be mistaken, but I am unable to properly view both the rdf triples table and the arsenic properties table (they overlap). I experience this both with firefox 2.0.0.3 and IE 6.0.2.
Thanks for your very interesting posts 🙂
(1)Sorry – this is a problem with WordPress. I will try to add some space
(2) should be better now. Possibly useful to have small type